PHYSICS | NECTA 2018 | Question 07
(a) What is meant by radioactive decay?
(b) A certain sample with half-life of 8 days contains 16 g of iodine 131.
(i)Write an expression to show the decay process of the sample.
(ii) Use an expression in (b) (i) to sketch the graph then estimate the mass of sample which will remain undecayed after 20 days.
(c) Describe the use of Geiger-Muller (G-M) tube in detecting nuclear radiations.
-
SECTION 01
(a) What is meant by radioactive decay?
Is the spontaneous disintegration of an atom by loss of sub atomic particles such as protons and neutrons which is associated with emission electromagnetic waves.
SECTION 02
(b) A certain sample with half-life of 8 days contains 16 g of iodine 131.
(i)Write an expression to show the decay process of the sample.Starting with the general equation of disintegration …
N = No (1/2)t/T
N – Number/Mass of atoms remained atoms at any instance
No – Initial Number/Mass of atoms at the beginning
t – Time taken from the beginning of the disintegration process
T – Time taken to reach the half-life of the material
Given No and T let us substitute to obtain a simpler expression
N = 16(1/2)t/8
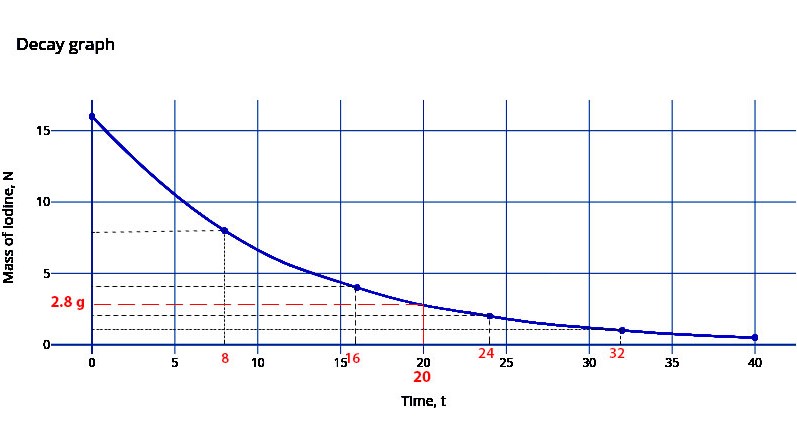
(ii) Use an expression in (b) (i) to sketch the graph then estimate the mass of sample which will remain undecayed after 20 days.
From the above equation we can obtain the following values if we intuitively substitute the multiples of 8, such as t=16, t= 24, t = 32, etc…
N = 16(1/2)t/8
at t = 0 days, N = No = 16g
at t = 8 days, N = 8g
at t = 16 days, N = 4g
at t = 24 days, N = 2g
at t = 32 days, N = 1g
Plot the graph …
SECTION 03
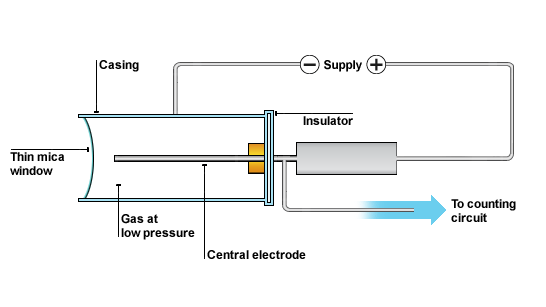
(c) Describe the use of Geiger-Muller (G-M) tube in detecting nuclear radiations.
The GM tube is a hollow cylinder filled with a gas at low pressure. The tube has a thin window made of mica at one end. The central electrode inside the GM tube is connected to high voltage across the casing of the tube as shown in the following diagram. When alpha, beta or gamma radiation enters the tube it produces ions in the gas as they collide with gas molecules. The ions created in the gas enable the tube to conduct for a particular short time interval and consequently produce voltage pulse. Each voltage pulse corresponds to single ionizing radiation entering the GM tube. The voltage pulse is amplified and counted on the display.
Geiger-Muller tube

More questions
Physics questions are much EASIER when you clear understand the concept. With Smart Darasa exprole concepts in fun and interactive way.
Learn more »Send Your Physics Question
Are you a STUDENT | PARENT | TEACHER, Send your physics question for FREE and we will get back to you with the solution or relavant link with similar question. We receive questions every day from 08:00hrs to 16:00 EAT. Depend on volume of questions, we send solutions the same day from 18:00hrs to 20:00hrs EAT or later next day.